Drs. Janet Mullington, Robert Thomas, Larissa Engert, Samuel Frank, Monika Haack, Jason Maley, Recep Ozdemir, Haoqi Sun, Alicia Stokes, John Torous, and Brandon M. Westover – Harvard Medical School and the Open Medicine Foundation-Supported Ronald G. Tompkins Harvard ME/CFS Collaboration
The goal of this project is to characterize several features of sleep regulation in ME/CFS and Long COVID compared with age and sex matched healthy sleep controls. The study will look at four primary components: 1) sleep-circadian dysfunction; 2) how immune function, specifically specialized pro-resolving mediators (SPMs), are changed in relation to sleep dysfunction; 3) brain electrical activity; and 4) mechanisms of state-boundary control and neurodegenerative diseases.
Dr. Wenzhong Xiao – Massachusetts General Hospital and the Open Medicine Foundation-Supported Computational Research Center for Complex Diseases
This project will use deep machine learning and network medicine to parse through clinical information, research findings, and multi-omics data of Long COVID, ME/CFS, and related diseases with the collective knowledge-base of drugs, diseases, genes, and symptoms to discover disease gene modules, and identify potential drugs as candidates for repurposing for these illnesses.
Prof. Alain Moreau – Université de Montréal and the Open Medicine Foundation-Supported ME/CFS Collaborative Center at CHU Sainte-Justine/Université de Montréal; Prof. Jonas Bergquist –the Open Medicine Foundation-Supported ME/CFS Collaboration at Uppsala University; Dr. Christopher Armstrong – Open Medicine Foundation-Supported Melbourne ME/CFS Collaboration; Dr. Wenzhong Xiao – Harvard University and the Open Medicine Foundation-Supported Computational Research Center for Complex Diseases; Dr. Dawei Li – Florida Atlantic University; and Dr. Tse Man Sze – Université de Montréal
This project hypothesizes that Long COVID, and ME/CFS after COVID, is the result of a broad molecular-level reorganization occurring primarily at the epigenetic level. The study aims to understand the molecular mechanisms underlying the development of long-term sequelae following a SARS-CoV-2 infection, by looking at 4 specific aims: 1) Global expression profiling of circulating microRNAs, 2) Global DNA methylation profiling, 3) Global proteomic plasma profiling, and 4) Global metabolomic plasma and urine profiling.

The Centers for Disease Control and Prevention (CDC) recognizes the significant overlap between Long COVID and Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS).
According to the CDC, Long COVID, also known as Post-Acute Sequelae of SARS-CoV-2 infection (PASC), can include a range of long-term symptoms that persist for weeks or even months after recovery from the acute phase of COVID-19. These symptoms can include fatigue, cognitive impairment (often referred to as “brain fog”), sleep disturbances, and other symptoms that are also common in ME/CFS.
The CDC acknowledges that further research is needed to understand the relationship between Long COVID and ME/CFS, including whether or not a subset of people with Long COVID are developing ME/CFS.
OMF is committed to leading this critical research, with the goal of improving diagnostic tools, finding effective treatments, and ultimately, discovering a cure for these complex diseases.
In March 2020, OMF launched an international, multi-year study across the six OMF supported Collaborative Research Centers (CRC). This was the first study of its kind and the aim is to examine the progression from COVID to Long COVID and the eventual progression to ME/CFS.
In March 2020, OMF brought together its global collaborative network, across 6 research centers in 4 countries, to carry out this global effort in several stages, using an initial $1,000,000 grant donated specifically for this effort. We are actively working to raise additional funding to continue this critical study.
Researchers are testing and analyzing data from individuals from the point of early COVID-19 illness through their recovery or prolonged illness state. This study has an opportunity to reveal what causes one to fully recover versus those at high risk of developing Long COVID and of those, the development of ME/CFS.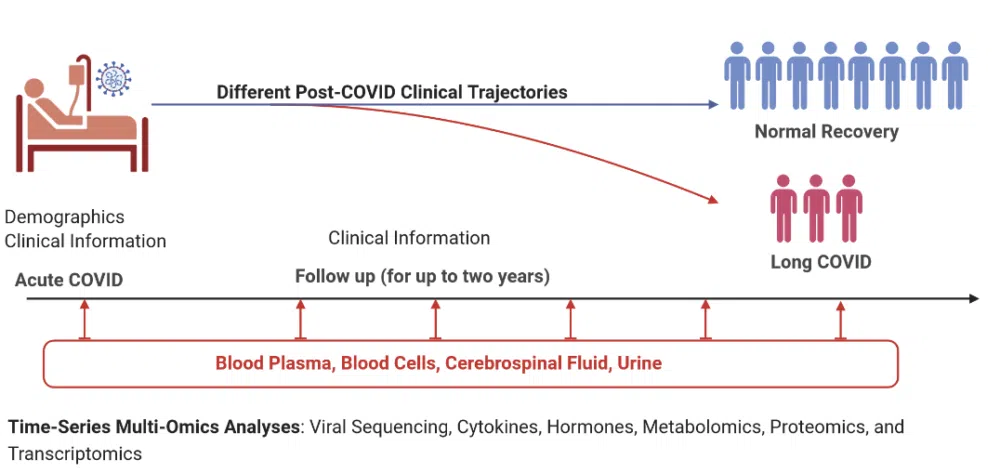
This data is also being compared to the OMF-Funded Post-Viral Complications in Herpes Simplex Encephalopathy (HSE) study data to provide another post-viral contrast. This study design offers the chance to elucidate precise pathological mechanisms of Long COVID, ME/CFS and other post-viral illnesses, identify potential diagnostic biomarkers and validate potential treatment targets for future clinical trials.

Updates from Phase 1
Updates from the Long Covid Clinic at Uppsala
Latest Updates from Montreal
Latest Updates from the Computation Center
What are the advantages of giving from your Donor Advised Fund (DAF)?
How do I make a donation through my DAF?
Just click on the DAF widget below. It is simple and convenient to find your fund among the over 900 funds in our system.
Still can’t find your fund?
Gifting of Stock
Broker: Schwab
DTC #: 0164
Account #: 47083887
Account Registered as:
Open Medicine Foundation
29302 Laro Drive
Agoura Hills, CA 91301
Please speak to your personal tax advisor and then email or call OMF at 650-242-8669 to notify us of your donation or with any questions.