by Cort Johnson
Autism is one of the most difficult conditions to treat. A neurodevelopmental disorder that typically strikes before the age of two, about a dozen different areas of the brain are affected.
Autism is characterized by social withdrawal, problems with both verbal and non-verbal communications and repetitive behaviors. Autistic infants tend to smile less and have less eye contact with others than normal, have trouble in social situations, and are less verbal. The prognosis for autistic children is bleak with one study finding that only 4% ever achieve full independence.
Autism Spectrum Disorder (ASD) and ME / CFS
ASD is not Chronic Fatigue Syndrome (ME / CFS) but some similarities exist. Both diseases affect cognition and sensory processing, cause problems with stimuli, cause significant social withdrawal, and are associated with increased levels of oxidative stress, reduced glutathione levels, and a Th2 immune response shift.
According to Dr. Naviaux, “ASD and ME / CFS are on the same biological spectrum.” Dr. Naviaux has encountered teenagers with ASD who develop ME / CFS, and adults with ME / CFS who develop autism-like symptoms of mutism, social withdrawal, sensory hypersensitivities, and OCD-like symptoms.
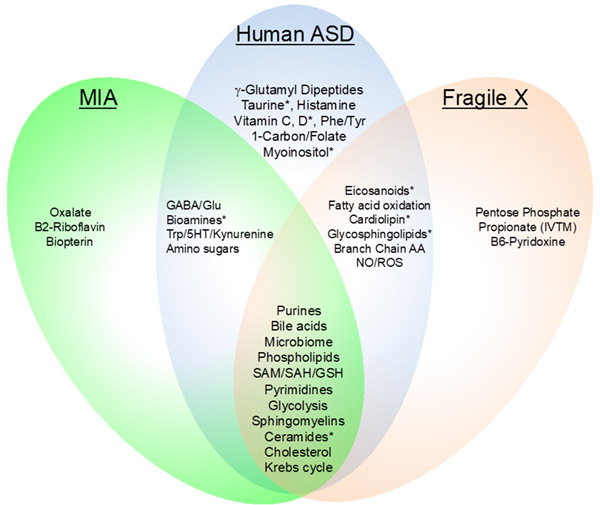
Both, he believes, are caused by a failure of the cell danger response (CDR) to shut down normally after a chemical or biological injury has been healed or cleared. Both disorders lead to abnormalities in metabolism that he has characterized using a laboratory tool called mass spectrometry and metabolomics. (The similarities in metabolism are illustrated in Figure 1.)
Some of the abnormalities shared by both disorders include disturbances in purines, sphingolipids (including sphingomyelins and ceramides), phospholipids, and the microbiome. Depending on whether a person is still actively fighting a perceived threat like a virus, bacterium, or toxin, or whether they are trying to heal after the exposure, these pathways can be increased or decreased compared to normal. The take-home message is that these are precisely the same pathways the cell uses normally for fight infection and recovery from injuries.
| A. ASD
|
B. ME / CFS
|
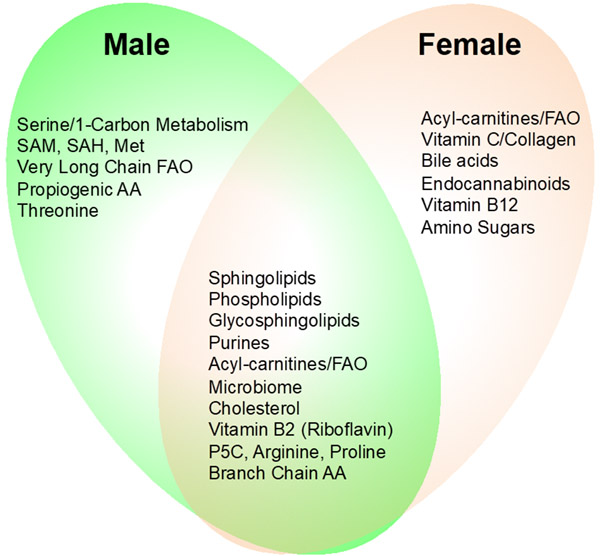
| Figure 1. Metabolic Features of Autism Spectrum Disorder (ASD) and Myalgic Encephalitis/ Chronic Fatigue Syndrome (ME / CFS). A, Males with ASD. MIA (maternal immune activation) and Fragile X are mouse models of ASD. The blue ellipse illustrates the metabolic pathways that were found to be normalized by suramin in children with ASD. (from Naviaux, et al., ACTN, 2017) B, Males and females with ME / CFS. Note that many pathway abnormalities in ASD are shared with ME / CFS. These include purines, sphingolipids (including sphingomyelins and ceramides), phospholipids, the microbiome, and cholesterol/sterol/bile acid metabolism. [from Naviaux, et al., PNAS, 2016) | |
Chronic Fatigue Syndrome (ME / CFS) is so similar metabolically to autism that Dr. Naviaux believes the same antipurinergic drug he’s trying in autism may be useful in ME / CFS – hence the interest in Dr. Naviaux’s autism trial.
Low-dose suramin in autism spectrum disorder: a small, phase I/II, randomized clinical trial Robert K. Naviaux, MD, PhD*1-4; Brooke Curtis, PsyD12; Kefeng Li, PhD1,2; Jane C. Naviaux, MD, PhD1,5; A. Taylor Bright, PhD1,2; Gail E. Reiner, DNP, FNP-C1,5; Marissa Westerfield, PhD6; Suzanne Goh, MD11; William A. Alaynick, PhD1,2; Lin Wang, MD, PhD1,2; Edmund V. Capparelli, PharmD3; Cynthia Adams, RPh7; Ji Sun, PharmD, PhD7; Sonia Jain, PhD8; Feng He, MS8; Deyna A. Arellano, BS7; Lisa E. Mash, BS6,10; Leanne Chukoskie, PhD6,9; Alan Lincoln, PhD12; Jeanne Townsend, PhD. Annals of Clinical and Translational Neurology.
The first thing to get about this study, Naviaux explains in the FAQ released with its publication, is that it’s not about the drug; it’s about why this drug – a very unusual one – appears to have worked. Suramin is an old anti-purinergic drug that’s been used, of all things, to fight African sleeping sickness.
Energy production – low metabolism – plays a huge role in Naviaux’s thought. Two key facts to remember regarding Naviaux’s hypothesis is that the mitochondria do more than produce energy and ATP does more than provide energy. Studies suggest that mitochondria’s remarkably fast metabolism allows them to respond more rapidly to a cellular event. Mitochondria are the first, for instance, to inform the innate immune system of danger. They also regulate cellular oxygen and hundreds of metabolic reactions.
Autism Spectrum Disorder: The Cell Danger Response

When a cell becomes too stressed it rather spectacularly releases its hard-won ATP out of the cell according to cellular stress levels. The ATP then binds to purinergic receptors on the cell – invoking the “cell danger response” – a metabolic reflex which puts the cell into “defense mode” by reducing its oxygen consumption, stiffening its membranes, releasing antiviral and anti-microbial factors, etc. The early steps of glycolysis, which metabolomic and mitochondrial studies keep highlighting in ME / CFS, are increased.
From hibernation to torpor or estivation to dauer, Naviaux is able to point to a variety of low energy states found in nature that may be used as defensive mechanisms.
In this capacity, suramin and other antipurinergic drugs, are a kind of molecular armistice therapy (MAT), signaling the cellular war is over, the danger has passed, and cells can return to “peacetime” jobs like normal neurodevelopment, growth, and healing. Naviaux FAQ
Naviaux put his cell danger hypothesis to the test with a small double-blinded, placebo controlled, pilot study to examine the safety and activity of low-dose suramin in one of the most treatment resistant diseases of all – autism spectrum disorder. It was a small study but a big step forward. Naviaux has used suramin to reverse autism-like behaviors in mouse models of autism but this was the first time he’s tried it in humans.
The Results
“We saw improvements in our son after suramin that we have never seen before”. A Parent
Naviaux moved cautiously giving five children just one dose to get an idea of the drug’s safety, efficacy and its effects on the children’s metabolomics.
It’s rare that drugs move the needle on autism but this one did. Remarkably 100% of the autistic children responded while none of the autistic children on the replacement drug did.
Six weeks after getting one dose of the drug, the average children’s autism (ADOS) scores dropped twenty percent from 8.6 to 7.0 – nearly removing them from an autism diagnosis. (Children with an ADOS score of 7-10 are considered to have autism). The ADOS scores of the five children getting the placebo drug, on the other hand, didn’t change at all. The scores indicated that the children’s social interactions, language, and restricted and/or repetitive behaviors all improved.
“New behaviors and new language was apparent every day.” A Parent
The statistics were promising but it was the parents stories of their children’s improvement that were most eye-catching.
Century-Old Drug Tested in Boys with Autism

The parents of two children reported that their children spoke the first sentences in their entire lives about a week after the dose. Two other children who had been non-verbal started making sounds for the first time in their lives and singing nonsense tunes. One 14-year old boy who had been silent for 12 years started speaking in sentences. For the first time ever he surfed on the computer and even discovered a 12 year old video he remembered before he became ill. At about 2.5 years he stopped developing normally. After suramin, he wanted to play hide-and-go-seek in the first week, tag in the next week, then soccer and games of catch in weeks 3 and 4. Suramin appeared to have removed a roadblock in development. Once removed, the normal sequence of development was able to start up again at an accelerated pace.
Another child improved so much that his teacher at school – unaware that he had tried suramin – sent a note home stating that he completed three weeks of schooling in three days.
Check out more parent stories of their children’s progress
The improvements increased for about three weeks and then declined. (Note that the testing was done at six weeks – after the improvement had begun to decline. The next study will include multiple doses.) A much larger study (40 or more participants) will be needed to get a more definitive answer but the trend was encouraging.
If you look up information on suramin’s safety you’ll find a raft of side effects but none were seen in Naviaux’s trial probably because of the low dose used. Naviaux explained that the vast majority of side effects seen with Suramin come when it’s used at cancer chemotherapy doses 25x’s higher than used in his study.
Conclusion
The suramin trial was a notable success. Despite the fact that only one dose was used, all of the five children with autism demonstrated significant improvements – a remarkable finding in a disease which has been so treatment resistant.
One of the most promising aspects of Naviaux’s cell danger hypothesis is that it suggests that a drug like suramin which resets the cell danger response would need to be taken just long enough for the cells to begin operating normally again. Next up, hopefully, then for autism and Dr. Naviaux, is a larger trial, and even more significant and long-lasting improvements.
Dr. Naviaux believes that the energy and other problems in Chronic Fatigue Syndrome (ME / CFS) may be caused by the same cell danger response he believes is present in autism. The strange wired and tired symptoms, the inability to settle down and rest, the hypersensitivities to stimuli, the ADHD-like problems, even the mood issues like anxiety – all are the consequence of the body being put in a low energy state. Ironically, people in low energy states just can’t relax because, Dr. Naviaux asserts, “it takes more energy to relax than react”.
Dr. Naviaux’s ME / CFS metabolomics study put the spotlight on that subject in ME / CFS. An expanded study is under way but a successful ME / CFS clinical trial would truly be the cat’s meow for this treatment resistant disease. Given that similar metabolic features have been found in both ME / CFS and autism, a suramin ME / CFS trial surely makes sense.
Publication:
Low-dose suramin in autism spectrum disorder: a small, phase I/II, randomized clinical trial. Naviaux et.al. May 2017 http://bit.ly/NaviauxASD
Q&A with Dr. Naviaux:
Dr. Naviaux answers questions about the SAT 1 Study.